Therapeutic Antibody Centre:
Antibodies and therapy
A hundred years of antibody therapy
In 1895 Hericourt and Richet described the first trials in which cancer cells were injected into animals to raise an antiserum for treating the patient. Their results seemed very promising and several patients with different types of advanced cancer were treated, each receiving an individual, tailor-made antiserum. None of them was cured, but they showed significant improvements in their symptoms. During the early 1900's many workers repeated these trials, but found inconsistent and contradictory results, leading to the conclusion in 1929 that "nothing may be hoped for at present in respect to a successful therapy from this direction".
 Click here to see how antibodies were predicted by Erlich 100 years ago.
Click here to see how antibodies were predicted by Erlich 100 years ago.
The problem was that the antisera were too variable, and the treatment was not reproducible and sometimes gave bad side-effects. It was discovered that an antiserum contains a mixture of many different antibodies, each one directed against a different antigen on the cancer cells. Many of these antigens are also present on normal tissue cells and so the antibodies against them could be harmful. In contrast, antisera against foreign substances like snake venom, did not have this problem and have always been very effective.
Monoclonal antibodies bring new hopes
In 1975 Kohler and Milstein found the key to aiming the "magic bullets" first envisaged 80 years earlier. Monoclonal antibodies, derived from the progeny of a single immune cell, were pure and available in potentially unlimited quantities. Since then a prodigious number of different monoclonal antibodies have been made and they have been exploited in almost every branch of biomedical research. Several have been found which recognise human cancers and some of these have been tested in clinical trials.
The first studies relied on the intrinsic ability of the antibody molecules to recruit the body‘s own defence mechanisms to kill tumour cells. When these natural mechanisms were found to be insufficient, various artificial "warheads" were proposed including conventional chemical drugs, radioisotopes and plant toxins. Now there are so many potential combinations of "guidance system" and "warhead" that we can test only a small fraction. However, we have learned some important lessons. Therapy with antibodies (and any other biological molecules) is limited by the ability of the patient‘s immune system to detect the new substance and neutralise it. Antibodies can be disguised as human to evade the immune response, but the "warheads", being foreign substances, are soon recognised. This means there is only a short time when antibody therapy can be used before it becomes ineffective. More fundamentally, the sheer numbers of tumour cells in patients with terminal disease overwhelm the therapy. Some cells express too little antigen to be targeted and cells in a large mass may escape attention altogether. Antibody therapy is most likely to be useful in the context of minimal, but disseminated disease; in fact just the situation that often occurs after conventional chemo-radiotherapy. This concept is not new; it was already foreseen by Hericourt and Richet in 1895. However, it is hard to tell whether the treatment of minimal disease is beneficial since it is difficult to measure an immediate effect. Instead we must wait to see whether treated patients survive longer than those who receive no treatment. Such trials take many years to complete and involve hundreds of patients.
Problems for commercial production
This is not a happy prospect for the pharmaceutical industry. The very specificity of antibody therapy guarantees that the market is small, because every different type of cancer requires a different product. Nevertheless the investment required to carry out clinical trials is enormous. The selection of product is difficult and critical; it is not sufficient to make a choice for partisan reasons like the ownership of patent rights. Instead it should be guided by scientific and clinical studies with as wide a variety of products as possible. A single antibody will probably not recognise all the tumour cells in a patient, but combinations of antibodies may be significantly more effective. These combinations need to be tested in small pilot studies using new methods for detecting and measuring small numbers of tumour cells to get an early indication of the likely outcome.
Passive therapy with antibodies
Other applications of monoclonal antibody therapy have already met with success; and this we can partly attribute to the use of antibodies in their natural role, which is to protect us against infection. For many years antisera from immunised animals or volunteer donors have been used to transfer immunity to patients who are vulnerable to infectious diseases. Now monoclonal antibodies are starting to replace them, offering substantial advantages in terms of potency, reproducibility and freedom from contaminants. The first to reach the market is palivizumab (Synagis), an antibody to respiratory syncitial virus (RSV), which can cause severe illness in newborn infants.
 Click here to see how human monoclonal antibodies are made
Click here to see how human monoclonal antibodies are made
Rhesus disease is a potentially fatal illness which affects unborn babies that carry the rhesus antigen on their red blood cells. If their mother lacks the antigen but has been immunised against it during a previous pregnancy, she can make antibodies which cross the placenta and destroy the red cells of the foetus. It is easily prevented by giving a small amount of anti-rhesus antiserum to the mother at the right time to prevent immunisation. However, suitable antisera are increasingly hard to obtain and the UK Blood Products laboratory has been testing monoclonal antibodies as replacements. They have carried out research to find those monoclonal antibodies, or combinations, which best reproduce the subtle effects of the natural antisera, and we see no reason to suppose that similar effort will not be equally rewarding in other situations where whole antisera were previously used.
New options for autoimmune disease
So far, the story of immune therapy has been a recapitulation of themes from the last century. It is in the areas of transplantation and autoimmune disease that recent discoveries in Immunology are having a new impact. We now know that T lymphocytes play a central role in initiating and controlling the immune response and we are unravelling the molecular basis for immune recognition and the way the body distinguishes invaders from its own tissues. This is bringing new insight into a wide range of severe diseases, including diabetes, rheumatoid arthritis, multiple sclerosis and many others, all of which appear to result in whole or in part from a breakdown of the normal mechanisms of self-tolerance. The damage which occurs in these autoimmune diseases is analogous to the rejection process which occurs after transplantation of a foreign organ.Normally the immune system learns to accept self tissues during early foetal development and this happens mainly by elimination of auto-reactive cells. However, adults also have active mechanisms for maintaining self-tolerance which are controlled by a complex symphony of interactions between different parts of the immune system. We can use antibodies, as well as other natural molecules, such as the hormone-like cytokines, to modulate the cellular orchestra and recreate the natural harmony. In animals it is possible to cure autoimmune disease and to prolong the survival of transplants indefinitely. The best results have been obtained by using combinations of agents working in synergy.
Our first efforts to translate these experimental results into human therapy were relatively crude, but met with some success. We used an antibody ("CAMPATH1H") which can destroy lymphocytes and disrupt the immune system at the most fundamental level. Unlike cancer therapy, we were not aiming to kill all the target cells, but just remove enough to allow a respite for the tissue under attack. The hope was that when the immune system regenerated over the subsequent months, the vicious cycle of tissue injury, resulting in fresh immune stimulation, would have subsided sufficiently for the natural control mechanisms to restore a healthy balance. This treatment was tested in patients with rheumatoid arthritis. Disease activity was reduced during the period of lymphocyte depletion, but in many patients it came back after three to six months when the lymphocytes started to regenerate. Experiments showed that we had to control the T cells during the period of regeneration in order to direct them into a more appropriate pathway of self-tolerance. To do this we need antibodies that block the function of critical molecules without killing the cells. One such molecule is the CD4 antigen; it is a vital component of the group of T lymphocytes which lead the immune orchestra. Antibodies against CD4 can divert the T cells from an aggressive to a tolerant state. This is being tested in the clinic at the present time. Another approach is the use of antibodies directed against the cytokine, tumour necrosis factor (TNF), which is a powerful stimulant of immune aggression. Anti-TNF agents have substantial activity in rheumatoid arthritis and several are now on the market (infliximab, etanercept, adalimumab). However, their effects are relatively short-lived and they must be administered repeatedly. The challenge is to find therapies, perhaps using combinations of these biological agents, which will give radical long-term effects.
Looking to the next century
Recent developments in immunology are opening up a huge range of potential new therapies. Other promising areas include vaccine development, recombinant or modified growth factors and their receptors, and the possibilities of therapy with whole cells. However, the very diversity and exquisite specificity of these new approaches pose huge new challenges to the pharmaceutical industry and drug regulatory mechanisms. Exploitation which is based mainly on market potential, intellectual property rights and dividend forecasts will produce a few commercially successful products, but will deny patients many of the possible benefits of decades of research. We expect that combinations of different biologicals may give beneficial effects beyond the mere sum of their components. This is not original; it has already been widely exploited in chemotherapy for cancer and antibiotic therapy for infectious diseases. The novel aspect of immune therapies is that they are based on the natural molecules which normally operate in concert to modulate and control the immune system. To get full benefit from our growing understanding of the immune system will require the same sort of open handed cooperation and collaboration in clinical research which has characterised experimental immunology during the past decades.
 Click here to show new ways to control the immune system
Click here to show new ways to control the immune system
Principles and Projects
The Therapeutic Antibody Centre (TAC) was set up by Geoff Hale and Herman Waldmann to provide a way of making a range of therapeutic monoclonal antibodies available for clinical trials in order to explore the basic principles of antibody therapy and provide the groundwork for later commercialization of useful products. In all, more than 1 kg of antibodies has been made and over 5000 patients treated; which is more than many substantial biotech companies can boast. Much of the early work was with CAMPATH1 antibodies, widely used for prevention of graft-versus-host disease and graft rejection in patients receiving bone marrow transplants and now licensed for treatment of leukaemia. Recent results in multiple sclerosis are particularly encouraging; patients have remained virtually free of new brain lesions for several years after a single treatment.An important principle of the TAC is the ability to take problems identified in the clinic back to the research lab in order to provoke a fresh cycle of research and development. This iterative process is now leading to a "second generation" of therapies which we believe will give us short-term treatments with a long-term impact.
The TAC team
The most important part of any organisation is the staff. Before Geoff Hale and Herman Waldmann founded the TAC in 1990, all the work of cell culture and antibody purification for clinical trials was done by just one person, Jenny Phillips, and she was only employed part-time! She was joined by Annamarie Drumm (Production Manager), Patrick Harrison (QA manager), and over the years in Cambridge they were assisted by Donna Stock, Julie Scanlan, Angela Shaw and Jeremy Holgate.When the TAC moved to Oxford, only Patrick was able to come and so a new team had to be built up again. Pru Bird was the first to join and she took on the task of transferring new antibodies from the research team and getting them ready for large scale production. A close knit and committed group was established over the subsequent years and remains the key to the TAC success.
Technology of Antibody Production
Most antibodies in the TAC are made from culture of mammalian cells in hollow fibre fermentors. At the centre of these machines is a kidney dialysis cartridge in which the cells can grow to a very high density, being perfused with culture medium through hundreds of fine fibres. The great advantage of this method is that a comparatively concentrated product can be obtained compared with more traditional techniques of cell culture. Before starting a production cycle, a large stock of frozen cells is grown from a single pure clone and carefully tested to check that it is free from any contamination by microbes or viruses.
Production starts when one of these vials is thawed and used to seed a small culture flask. The culture is expanded until there are sufficient cells to seed a fermentor and then the cells can be maintained continuously for three to nine months. At this stage large amounts of nutrient culture medium are required and the TAC pioneered the use of large (200 litre) plastic bags for holding the medium which is prepared and sterilised on site. The harvested antibody is filtered and stored frozen until the end of a culture run when it is pooled and purified by column chromatography. Usually three columns which exploit different properties of the antibodies are used; Protein A (affinity), S-Sepharose (ion exchange) and Superdex-200 (size exclusion). Together they give a very high degree of purification and contaminants are reduced to very low levels. In addition to numerous checks during the process, the final batches of antibody are subjected to a large panel of tests to ensure that they are functionally active and free from undesirable contaminants. The antibodies are stored frozen and distributed to collaborating physicians when they are requested for particular patients.
Clinical results from the TAC
The TAC has enjoyed the collaboration of many clinical colleagues around the world who provided opportunities to try out new ideas. Overall the clinical results have been better than could have been expected.
Bone Marrow Transplantation
The largest application of TAC antibodies was in bone marrow transplantation. Originally, CAMPATH1M was used to remove donor T cells and prevent graft-versus-host disease. This was one of the first clinical applications of monoclonal antibodies and it has been very effective. However, some of the undoubted benefit of this manoeuvre was offset by an increase in graft rejection and, in some cases, of leukaemia relapse. Subsequent studies used CAMPATH1G to deplete host T cells in vivo to address these problems. The new protocols showed clear improvements over the previous results. When we started this programme, the problems of GvHD and rejection meant that it was usually only possible to transplant bone marrow between HLA-matched siblings. This restricted the use of marrow transplantation to about a quarter of patients who might benefit. Now, with the establishment of large panels of tissue-type volunteers, the availability of antibodies for controlling transplant complications has led to a great increase in the number and success rate of transplants from unrelated donors.
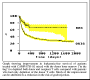 Click here for some results from bone marrow transplants
Click here for some results from bone marrow transplants
An even simpler method for controlling both GvHD and graft rejection was pioneered by colleagues in Israel, South Africa and Holland. CAMPATH-1G or CAMPATH-1H is added directly to the bone marrow before infusion. It can then deplete both the donor and the recipient T cells at the same time. The results of these trials have been particularly good and the procedure has been extened to the treatment of peripheral blood stem cells - which provide an alternative to bone marrow for transplantation that is much less uncomfortable for the donor. More recently, other developments allowed stem cell transplants to be used for older patients, when CAMPATH-1H was introduced as a key component of reduced intensity conditioning.
Leukaemia and lymphoma
Early on in our work, we used several monoclonal antibodies for the direct treatment of leukaemia and lymphoma. The best results were with CAMPATH1G and CAMPATH1H, and some good remissions were obtained in patients who were unresponsive to conventional drugs. This work was taken up by The Wellcome Foundation but after several years they abandoned it because therapy did not prove to be so effective in patients with bulky disease in lymph nodes. However, it was spectacularly successful in patients with the rare prolymphocytic leukaemia and results in patients with the more common chronic lymphocytic leukaemia were also very good.
 Click here and see a treatment of leukaemia with CAMPATH-1H
Click here and see a treatment of leukaemia with CAMPATH-1H
Eventually the TAC found another company to develop CAMPATH-1H, which ultimately led to a product being approved for marketing in 2001. You can read more of this story in our article on the history of Campath .
Rheumatoid arthritis and vasculitis
Clinical trials in autoimmune diseases were pioneered at Addenbrooke‘s hospital, Cambridge in the departments of Rheumatology, Medicine and Neurology. Several antibodies have been tested, including CD25 (specific for activated T cells), CAMPATH1H (for depleting lymphocytes), CD4 (for blocking the function of the critical helper T cells) and CD18 (which blocks the migration of leukocytes from the blood to sites of inflammation).Treatment with CAMPATH1H alone gave good results in rheumatoid arthritis. Most patients entered remission or stable disease, and the durations of the remissions have varied from a few weeks to several years. Repeated treatment with CAMPATH1H is possible and effective, but some patients eventually made an anti-idiotype response against the humanised monoclonal antibody. However, the depletion was so effective that T cells do not return to normal levels for a very long time.
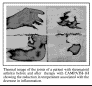 Click here for an example of arthritis treatment
Click here for an example of arthritis treatment
A new antibody-like substance was made in the TAC as an alternative treatment for rheumatoid arthritis and similar diseases. This is TNF receptor-Ig. It consists of the soluble portion of the membrane receptor for tumour necrosis factor linked to the Fc domain of a human IgG1 antibody. The chimeric construct had a very high affinity for TNF and was able too block this critical cytokine in vivo. The role of anti-TNF agents is now widely recognised and several are on the market.
Vasculitis is the term for a group of severe autoimmune diseases characterised by damage to blood vessel walls and a variety of symptoms in many different organs. Treatment with CAMPATH-1H to deplete lymphocytes has been effective both in patients with primarily cell-mediated disease, and somewhat to our surprise, it has also worked very well in patients with autoantibodies, eg Wegener‘s granulomatosis. This work was pioneered by the late Dr Martin Lockwood who made a huge contribution to the clinical development of monoclonal antibodies. He is much missed by patients and colleagues.
Multiple Sclerosis
Some of our most exciting results have been in the treatment of multiple sclerosis. In an initial study, more than 60 patients have been treated by colleagues at Addenbrooke's Hospital in Cambridge, with some very impressive results. After five days of treatment with CAMPATH-1H, the occurrence of new brain lesions was greatly reduced for many years. This has now progressed to a phase III clinical trial (sponsored by Genzyme Inc.) comparing two short courses of CAMPATH-1H a year apart with standard beta-interferon injections in relasping remitting MS patients. Analysis of the results at 3 years indicated that CAMPATH-1H treatment reduced the risk of accumulating disability by 70% compared to beta inteferon treatment.
Diabetes
A current phase 3 clinical trial (sponsored by ToleRx Inc.) is studying the effects of an aglycosyl CD3 antibody (Otelixizumab) for the treatment of Type I diabetes. It has already shown effiacy in the treatment of rheumatoid arthritis and kidney transplant rejection. Our hope in the diabetes study, is that by treating patients very early after diagnosis, it may be possible to arrest the process of destruction of insulin-producing cells and thereby enhance the subsequently quality of their life. The results of the phase I/II trial showed that the insulin requirments could be significantly reduced for at least 18 months after a single 1 week course of ChaglyCD3 antibody treatment, particularly in young, new onset type 1 diabetes patients with residual islet function at the time of initial treatment.
 Click here to show how antibodies may be helpful in new onset, type 1 diabetes
Click here to show how antibodies may be helpful in new onset, type 1 diabetes
Acknowledgements
This leaflet was written and produced by Geoff Hale and updated by Steve Cobbold. Our work has been funded by many sponsors over the years, including the Medical Research Council, Wellcome Foundation, The Gilman Trust, the Kay Kendall Leukaemia Fund, LeukoSite Inc, Millennium Pharmaceuticals Inc, TolerRx Inc, GlycArt Biotechnology AG, The EP Abrahams Trust, Cancer Research UK, The Leukaemia Research Fund. Thanks are due to literally hundreds of scientists, technicians, nurses and physicians who have produced the antibodies and used them in the treatment of patients.
This document was converted to HTML 13th May 1996 and most recently updated 5th March 2010.
This page was an HMS Beagle (BioMedNet)
 Web Pick and was given a
Web Pick and was given a  BioMedLink Award from 'the Yahoo for biological and medical researchers' .
BioMedLink Award from 'the Yahoo for biological and medical researchers' .

"Our aim is to provide the best antibodies for academic research"
Related topics:
Last Updated 5th March 2010 by Steve Cobbold
 Click here to show how antibodies can be effective in treating relapsing/remitting Multiple Sclerosis
Click here to show how antibodies can be effective in treating relapsing/remitting Multiple Sclerosis