The apparent lack of correlation between the number of genes in an organism and its resulting biological complexity raises interesting and fundamental issues in biology.
One possible source of our complexity is post-translational modification (PTM): the alteration of proteins after translation, typically through changes to their amino acid side chains. However, the processes that give rise to PTMs are complex and not template driven and often give rise to product mixtures. The result is that dissecting the effect of PTMs on protein function is challenging.
We have proposed [pub 49][pub 96] that one strategy for dissecting the structure-activity relationships of PTMs might be through the use of chemistry, through the construction of synthetic proteins containing PTMs or PTM-mimics.
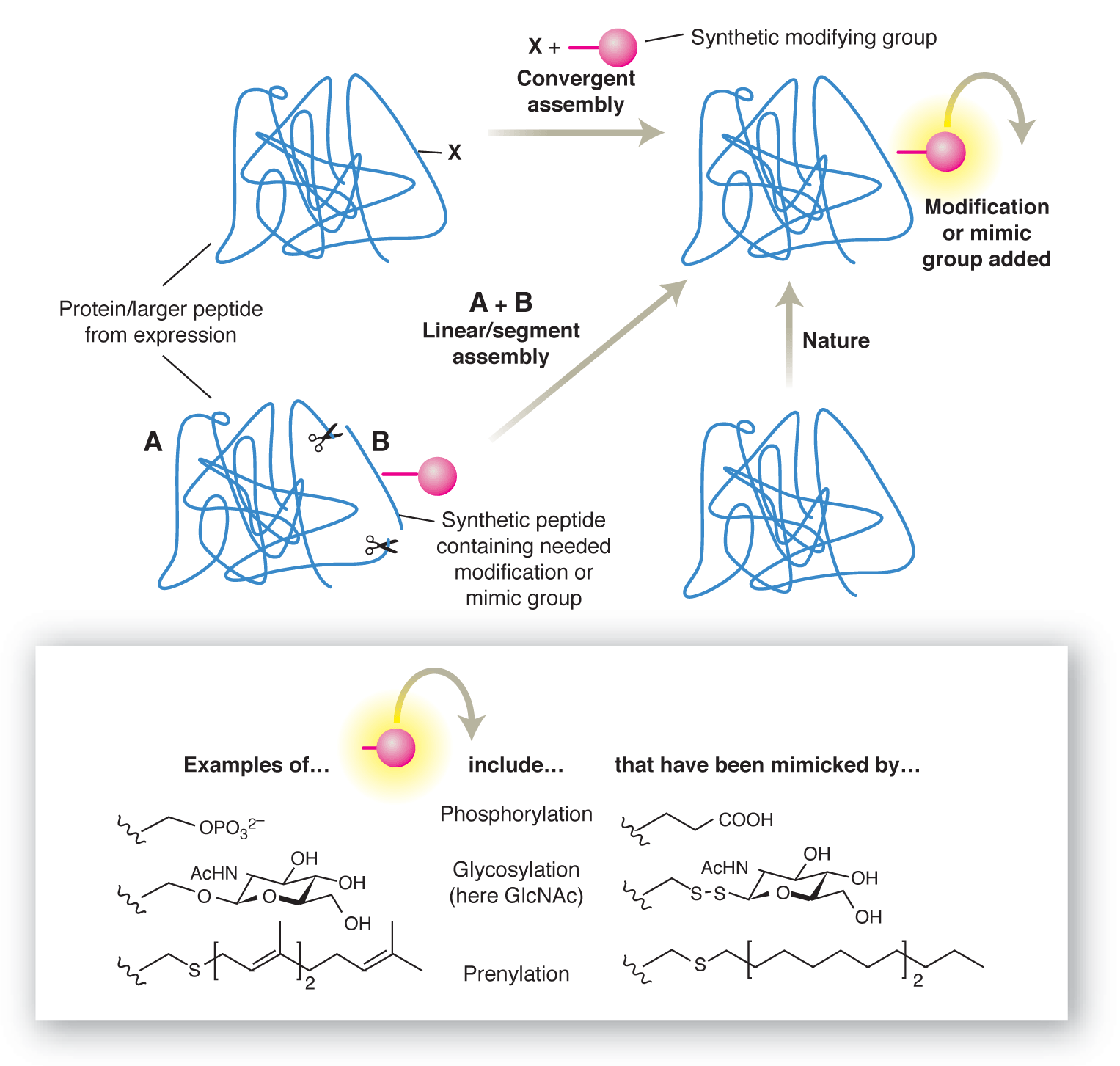
We have developed new tools [pub 96] that rely upon a "tag-modify" approach to introduce a wide range of modifications including glycosylation [pub 48][pub 57][pub 81][pub 85][pub 94][pub 97]; sulfation [pub 85]; prenylation [pub 97]; phosphorylation [pub 97] and methylation.[pub 97]
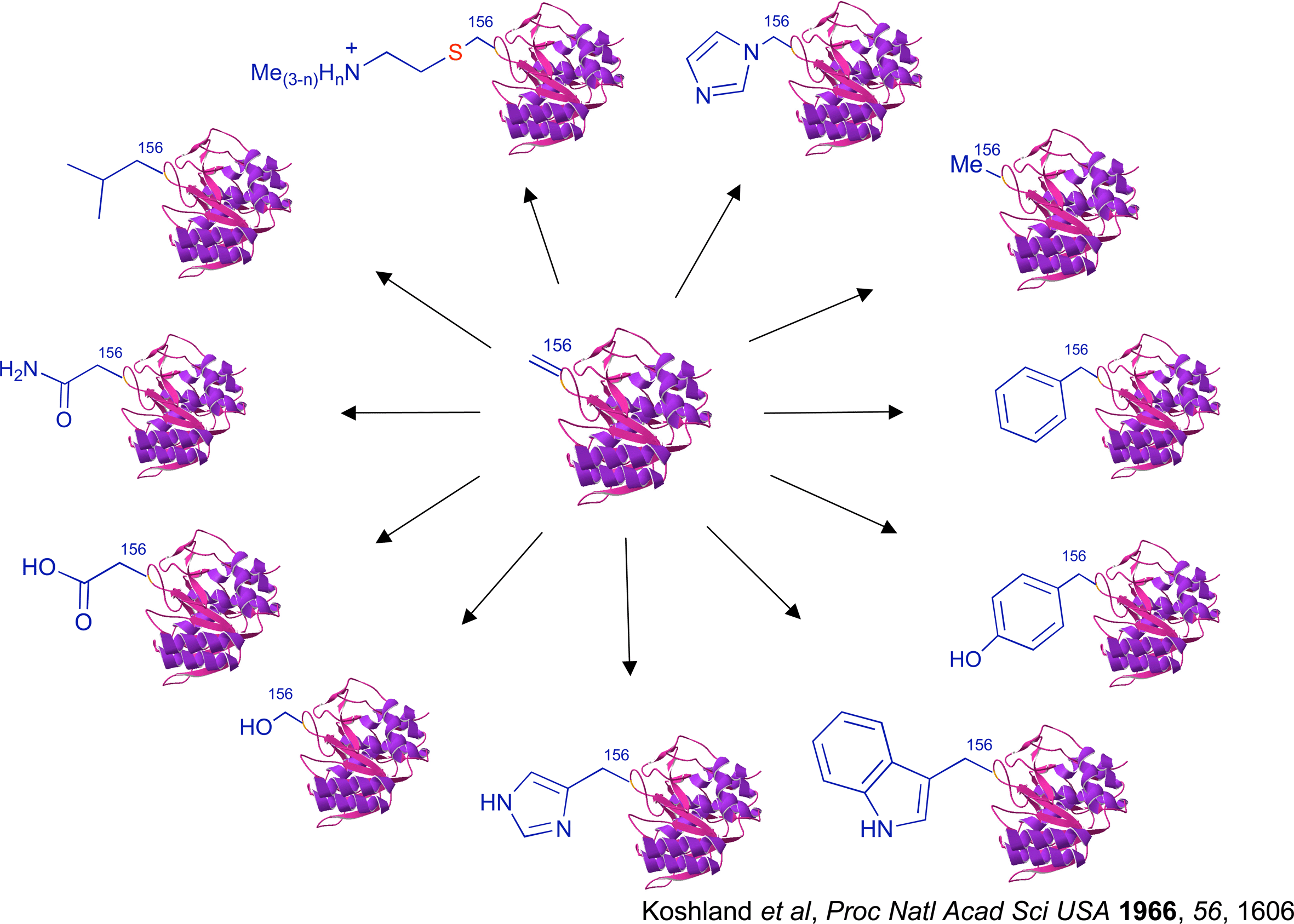
This work is now developing into broad strategies for accessing, through chemistry, proteins that have precisely altered residue structure. Chemical switching in proteins is allowing us to move towards the goal of "chemical mutagenesis".
Invention and use of dual tag-modify strategy has allowed us to build synthetic proteins that function as effective mimics both in vitro and in vivo. We have used these to map cognate binding partners in mammalian brain. [pub 85]
At the heart of our work is the development of synthetic methodology for chemoselective and regioselective manipulation of protein structure. These macromolecules are challenging and exciting starting materials for synthesis that must be manipulated with minimal protecting group strategy, so often relied upon in synthesis. This necessitates novel strategies [pub 85], novel reagents [pub 97], and novel catalysts.
Multicomponent & array chemistry[pub 58]has allowed us to assemble libraries of cell-permeable compounds that can modulate, interrupt and augment the processes that control protein modification
This strategy of is of particular relevance to PTM control in the absence of ready, effective genetic or silencing methods.
- Biomolecule & Bioconjugate Construction
- Probes of Mechanism in Biology
- Exploitation in Medicine & Biotechnology
- Chemistry for Biology
- Biology for Chemistry
- Exploring, Exploiting and Designing Proteins
- Synthetic Biology
- Post-translational Modification
- New Tools for Chemical Medicine
Prof Benjamin G. Davis
University of Oxford
Chemistry Research Laboratory
Mansfield Road
Oxford, OX1 3TA, UK
Phone: + 44 (0)1865 275652
Fax: + 44 (0)1865 275674
Ben.Davis@chem.ox.ac.uk
