Research
| Jarvis Lab |
Research |
||
|
Our research is focused on the biogenesis and operation
of chloroplasts (and other plastids) in plants. Particular areas of interest are the import
into chloroplasts of nucleus-encoded proteins, and ubiquitin-dependent proteolysis via a
process called chloroplast-associated protein degradation (CHLORAD). |

|
|
| Chloroplast Protein Import | ||
|
Plastids are a diverse family of plant organelles with numerous functions that are vital for plant growth. The family includes chloroplasts (which are responsible for photosynthesis), and a range of non-photosynthetic variants such as carotenoid-rich chromoplasts in flowers and fruits, starch-containing amyloplasts in seeds, tubers and roots, and chloroplast-precursor organelles in dark-grown plants called etioplasts (figure 1) [1]. Most plastid proteins are encoded by the nuclear genome and synthesized in the cytosol as precursors with N-terminal targeting peptides. Import of precursors into chloroplasts is mediated by multiprotein machines in the surrounding envelope membranes, called TOC and TIC (for Translocon at the Outer / Inner envelope membrane of Chloroplasts) (detail) [2,3]. Our research seeks to understand these import systems mechanistically, as well as their regulation through proteolysis. We have brought to bear the unique advantages offered by the model plant Arabidopsis thaliana (thale cress) as an experimental system in this field [4]. In parallel with our fundamental research using the Arabidopsis model, we also employ a range of crop species to investigate potential applications of the research. In doing these things, we apply a full spectrum of molecular, genetic, cellular and biochemical approaches. |
||
| Chloroplast-Associated Protein Degradation (CHLORAD) | ||
|
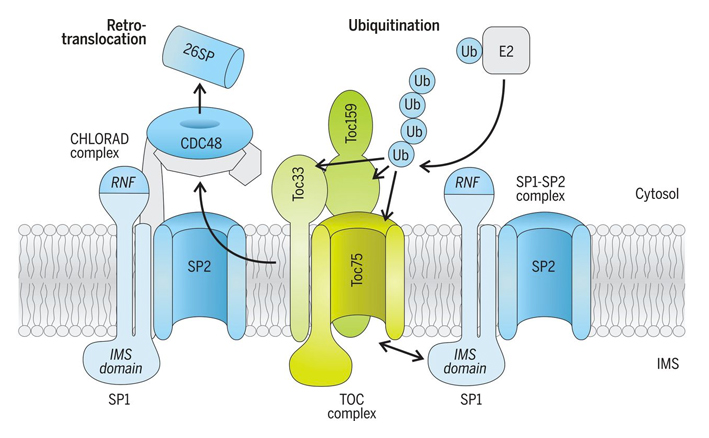
An especially significant breakthrough arising from our work was the discovery that plastids are directly regulated by the ubiquitin-proteasome system – thereby defining a new and fundamentally important field of cell biology [1,3]. Originally we identified a ubiquitin E3 ligase located in the plastid outer envelope membrane called SP1 (SUPPRESSOR OF PPI1 LOCUS1) (figure 7), and showed that this E3 ligase targets the protein import machinery (the TOC apparatus) for degradation [5]. More recently, we revealed that SP1 is part of a broader system, which we named CHLORAD; and showed that this system incorporates a protein "retrotranslocation" process that delivers target proteins to the cytosol, to enable their degradation by the cytosolic proteasome (figure 9) [6]. By regulating the protein import machinery in this manner, CHLORAD exerts considerable influence – it controls the plastid's proteome, functions and developmental fate (e.g., what plastid type is formed), which in turn affect plant growth and development more broadly [5,6]. Nevertheless, there are many open questions concerning the mechanistic details of CHLORAD, and addressing these questions is a major focus of our work. Because CHLORAD is important during those developmental transitions in which plastids change type (figure 1), its discovery suggested novel crop improvement strategies [5,6]. Indeed, it potentially has diverse agricultural applications; for example, during fruit ripening in crops like tomato, when chloroplasts transform into chromoplasts, or during grain development in crops like wheat and rice, when amyloplasts are formed. By manipulating CHLORAD activity, it may be possible to control such processes in a beneficial way, to improve the quality or yield of crops [7,8]. Our work revealed an important role for CHLORAD in plant abiotic stress responses [9,10]. Thus, manipulating CHLORAD may also enable the development of more stress-tolerant crops, which is a particular priority in the context of climate change. We are exploring these applied avenues in parallel with our fundamental studies on the molecular mechanisms of CHLORAD. |
||
|
References 1. Jarvis, P. and López-Juez, E. (2013) Biogenesis and homeostasis of chloroplasts and other plastids. Nat. Rev. Mol. Cell Biol. 14: 787-802. 2. Jarvis, P. (2008) Targeting of nucleus-encoded proteins to chloroplasts in plants (Tansley Review). New Phytol. 179: 257-285. 3. Sun, Y. and Jarvis, R.P. (2023) Chloroplast proteostasis: import, sorting, ubiquitination, and proteolysis. Annu. Rev. Plant Biol. 74: 259-283. 4. Jarvis, R.P., ed. (2011) Chloroplast Research in Arabidopsis: Methods and Protocols, Vols. I and II. Methods in Molecular Biology, Vols. 774 and 775. Humana Press, Totowa, NJ, USA. 5. Ling, Q., Huang, W., Baldwin, A. and Jarvis, P. (2012) Chloroplast biogenesis is regulated by direct action of the ubiquitin-proteasome system. Science 338: 655-659. 6. Ling, Q., Broad, W., Trösch, R., Töpel, M., Demiral Sert, T., Lymperopoulos, P., Baldwin, A. and Jarvis, R.P. (2019) Ubiquitin-dependent chloroplast-associated protein degradation in plants. Science 363: eaav4467. 7. Ling, Q., Sadali, N.M., Soufi, Z., Zhou, Y. Huang, B., Zeng, Y., Rodriguez-Concepcion, M. and Jarvis, R.P. (2021) The chloroplast-associated protein degradation pathway controls chromoplast development and fruit ripening in tomato. Nat. Plants 7: 655-666. 8. https://www.oxfordsparks.ox.ac.uk/videos/changing-plant-chloroplasts-to-improve-crop-performance/ 9. Ling, Q. and Jarvis, P. (2015) Regulation of chloroplast protein import by the ubiquitin E3 ligase SP1 is important for stress tolerance in plants. Curr. Biol. 25: 2527-2534. 10. https://www.oxfordsparks.ox.ac.uk/videos/hardy-crops-to-tackle-food-insecurity/ |
|
|
Last updated: Dec 2023
Paul Jarvis