
 |
| Home | Research | Publications | People | Joining the Group | Teaching |
Complex Cation-Ordered Oxide PhasesComplex metal oxides which exhibit ordered arrangements of cations often exhibit physical behaviour which is distinct from cation disordered analogues. However, the preparation of cation ordered phases is challenging as entropy strongly disfavours the formation of cation ordered materials at the high synthesis temperatures generally required for the formation of complex oxides. Recently we have demonstrated that by exploiting the differing coordination preferences of different cations, cation ordered perovskite phases can be prepared by introducing large numbers of anion vacancies into the extened framework of phases. |
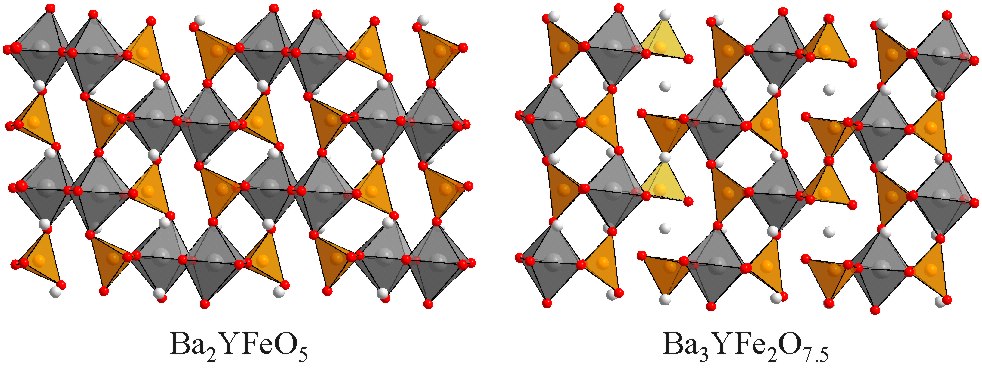 |
Cation Ordered Perovskite Phases Ba2YFeO5 and Ba3YFe2O7.5 adopt complex cation-ordered perovskite structures. The combination of a high concentration of anion vacancies and a large mismatch in the ionic radii of Fe3+ and Y3+ leads to highly unusual cation-ordered and anion-vacancy ordered structures being adopted by these phases in order to retain the local YO6 and FeO4 coordinations of the metal centers while minimizing lattice strain. These anion deficient phases can be topochemically oxidized as described here. Inorganic Chemistry 51 (2012) 12281. |
|
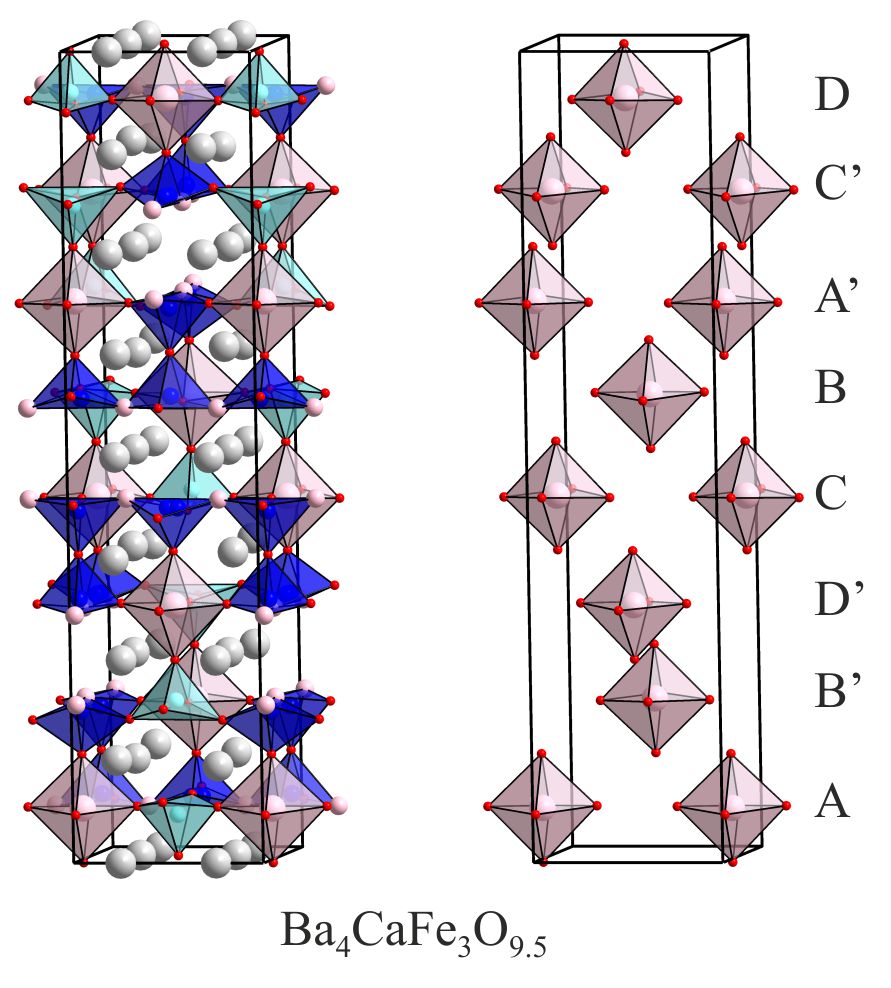
Non-centrosymmetric Materials via Cation Order Materials with non-centrosymmetric structures (those without inversion symmetry) can exhibit physical properties forbidden to centrosymmetric materials, specifically pyroelectricity, ferroelectricity, piezoelectricity and second-harmonic generation. Non-centrosymmetric materials are extremely rare due to the energetic favourability of highly symmetric packing schemes in solids. However, complex cation order can break the inversion symmetry of the host lattice in which the cations reside. For example the elaborate iron-calcium order induced into Ba4CaFe3O9.5 by the presence of large numbers of anion vacancies lifts the inversion symmetry of the host perovskite lattice allowing the material to exhibit piezoelectricity and second-harmonic generation. This method of utilizing cation order to break inversion symmetry in complex oxides offers a route to the preparation of multiferroic materials those which combine ferroelectric and magnetic behaviours. Chemistry of Materials 22 (2010) 5580. |
 |
 |
Cation Ordered Brownmillertie Phases The A2B2O5 brownmillerite structure is one of the most common anion-vacancy ordered perovskite structures adopted by complex metal oxides. It consists of sheets of apex-linked BO6 octahedra inter-leaved with sheets of BO4 tetrahedral coordination sites. By utilizing the relative preferences of different cations to adopt octahedral or tetrahedral coordination, cation-ordered A2BBO5 brownmillerite phases can be prepared such as Ca2Cr0.5Ga1.5O5, and the structurally related LaCa2Fe2GaO8. There is growing interest in this type of material as they have potential to act as efficient oxide ion conductors in high-temperature oxide fuel cells. Journal of Solid State Chemisty, 222 (2015) 71. |
|
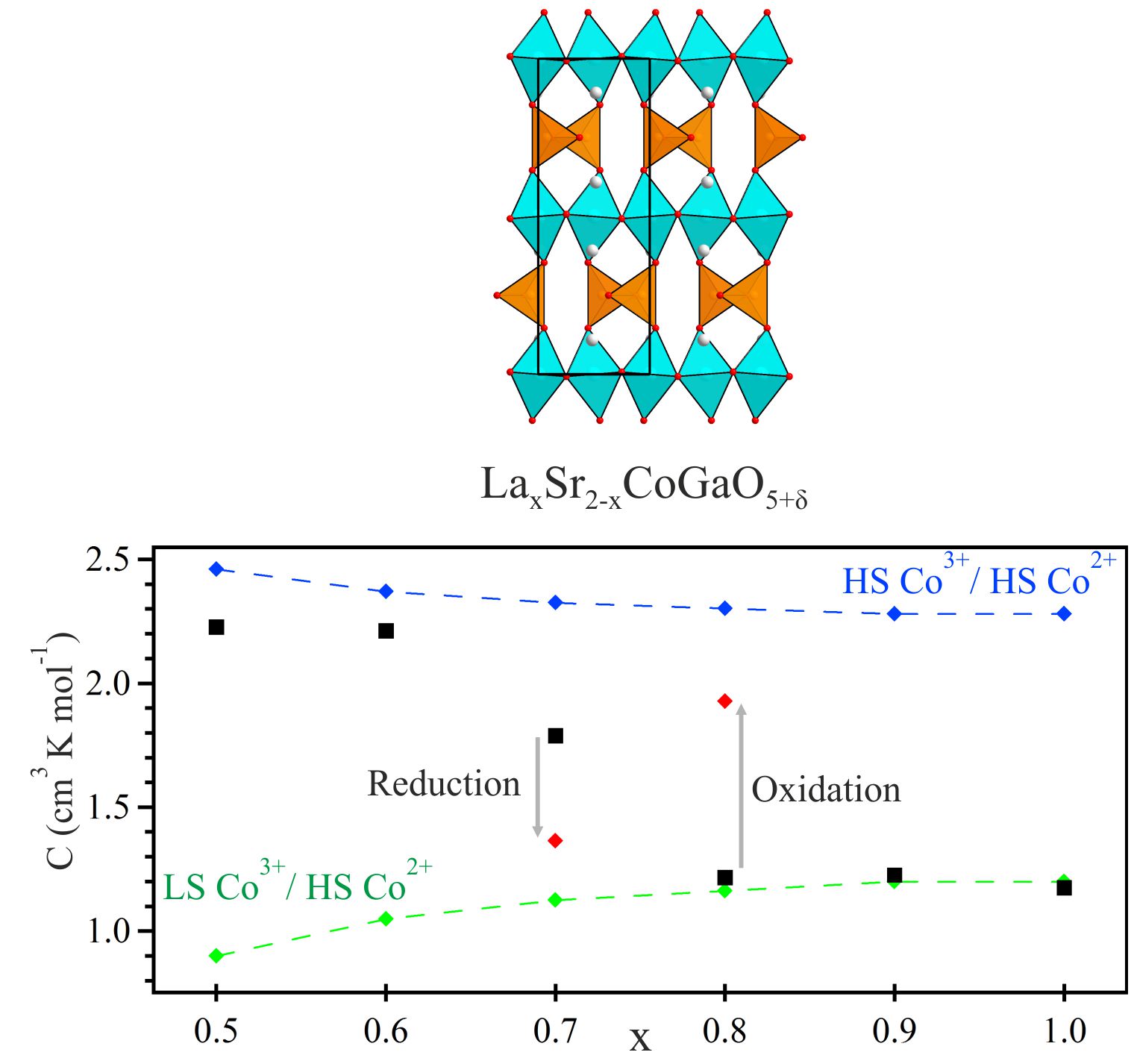
Oxygen-Sensitive Spin-State Transition Phases in the composition range LaxSr2-xCoGaO5+d (0.5 < x < 1) adopt anion-vacancy ordered brownmillerite-type structures. The aliovalent La3+/Sr2+ solid solution on the A-cation site of these phases, in combination with oxygen non-stoichiometry, results in a mixed Co2+/Co3+ oxidation state which evolves across the series from La0.5Sr1.5CoGaO5.01 (Co+2.52) to LaSrCoGaO5.18 (Co+2.36). Magnetization data indicate the Co3+ centers present undergo a change in spin-state, from high-spin, S = 2 to low-spin, S = 0, as a function of declining average cobalt oxidation state. The oxidation state dependence of the Co3+ spin-state is demonstrated by tuning the oxygen content of phases, demonstrating the potential of these materials in oxygen sensing applications. Dalton Transactions, 43 (2014) 1571. |
 |