Organometallic Research
The O’Hare group is interested in synthesising novel organometallic compounds and has recently concentrated on those containing the permethylpentalene (Pn*) ligand.
Once organometallic species have been prepared, the group explores the electronic and structural properties of these materials via cyclic voltammetry and crystallography.
A high yielding, facile synthesis of permethylpentalene was discovered by the group in 2007. Since then the group has utilised this ligand to form a series of varied organometallic complexes, a selection of which are reviewed below.
Arguably the most interesting development in Pn* chemistry was the synthesis of a series of double metallocenes. Single metallocenes, where a metal atom is sandwiched between two cyclopentadienyl (Cp) ligands have been known for some time, and now this group have utilised the Pn* ligand to form a series of double metallocenes.
The interplay between the atomic size and bonding requirements in these Pn* sandwiches gave a series of fascinating properties and interesting structural differences.
| V2Pn*2 | Cr2Pn*2 |
Claret-red crystalline solid Shortest organometallic V-V bond to date (2.169) 2 singlets in 1H-NMR (2:1 ratio) implies a D2h symmetry in solution |
Strong temperature dependence in NMR |
| Mn2Pn*2 | Co2Pn*2 |
Red-brown crystalline solid 2nd shortest Mn-Mn bond ever reported Paramagnetic (S=1) therefore difficult to characterise via NMR |
Centrosymmetric molecule, η5:η3-Pn* bonding mode 1H-NMR shows two peaks (1:2 intensity ratio) implying D2h symmetry in solution. Lack of Co-Co interactions contradicts predictions by DFT!
|
| Ni2Pn*2 | Fe2Pn*2 |
η3:η3-Pn* bonding mode 16e- Ni centres parallels stability of trans-[Ni(η3-allyl)]2(µ:η3,η3-Pn) NMR shows 2 singlets implying D2h symmetry |
Fe(II) acts as an oxidant and the anti-aromatic Pn* dimerises DFT has suggested that Fe2Pn*2may be a transition state to Fe loss. |
Other, more recent, work has been investigating the interesting η8-Pn* bonding mode, where the Pn* can wrap around the central metal atom. Specifically we are looking at the use of the Pn* ligand with the early transition metals
Recent Publications:
We have synthesised and structurally characterised group 4 metallocenes based on methylated indenyl rings. A benzyl analogue has been synthesised and along with other zirconocenes, has been tested for its activity as an ethylene polymerisation catalyst in solution. Synthesised complexes have been shown to outperform dichloride analogues by a factor of two.
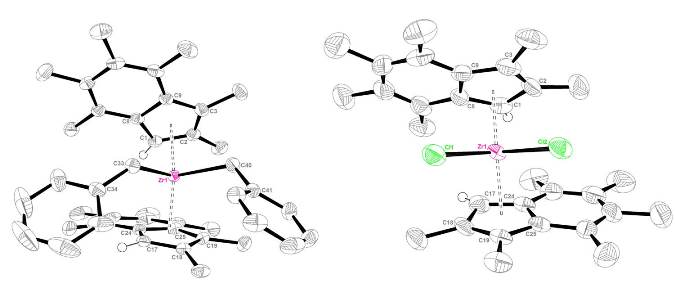
Complexes have been analysed by DFT and structural features of the compounds have been studied.
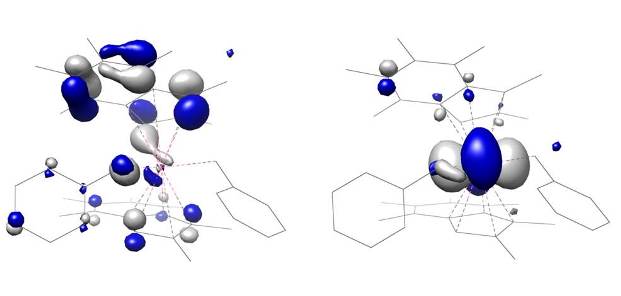
Research into related indenyl-based metallocenes and their use in polymerisation is ongoing.
Recent Publications:
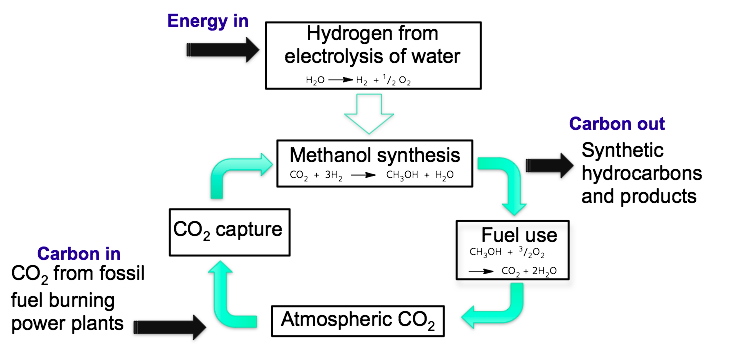
The role of carbon dioxide as a greenhouse gas and its contribution to global warming is widely recognised by both scientists and governmental agencies. It is now imperative that new reactions and processes are discovered that can either efficiently store or utilise the abundant and renewable CO2 resource in an environmentally friendly manner. However, we face a fundamental challenge because carbon dioxide is so kinetically and thermodynamically stable We have recently reported the first example of the selective hydrogenation of CO2 to CH3OH, using an FLP-based non-metal protocol at low pressures (1-2 atm). Current investigations are focussed on increasing the stability of the system towards hydroxylic agents, and hopefully thereby rendering the system catalytic.
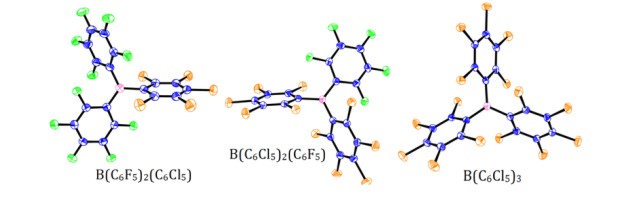
Due to the instability of B(C6F5)3, focus was turned to producing a hydrolytically stable Lewis acid which can be used in an FLP system. Three novel Lewis acids, B(C6F5)3-x(C6Cl5)x x = 1, 2, 3, were synthesised.
Current investigations are focused on finding a catalytic system which can reduce CO2 to methanol.
Recent Publications: